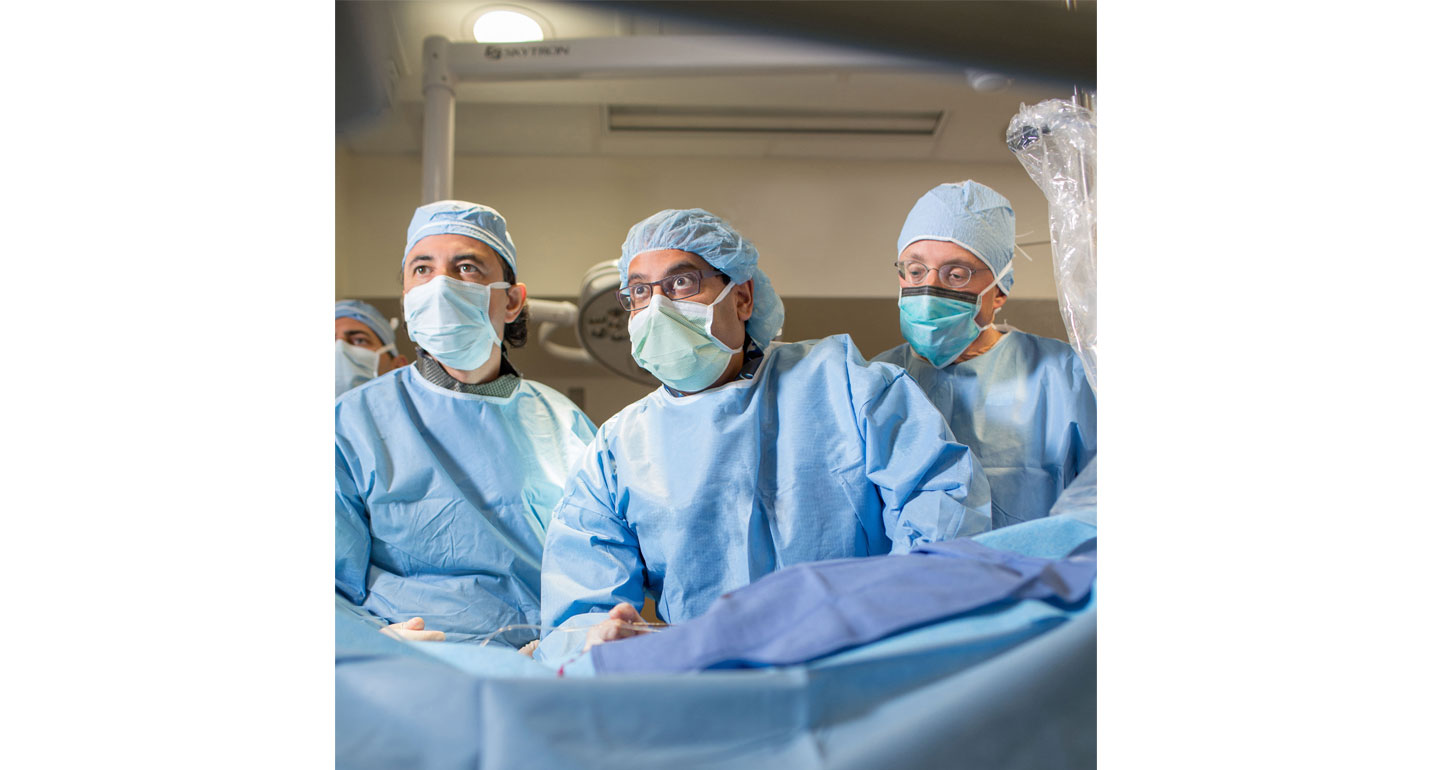
Next-generation permanent heart implant is the only FDA-approved device to reduce stroke risk in patients with atrial fibrillation
WASHINGTON — MedStar Health physicians have performed the 700th implant of the WATCHMAN™ device to reduce stroke risk in patients with atrial fibrillation, or AFib. MedStar Health is the first healthcare system in the Greater Washington-Baltimore region to achieve this milestone. Both MedStar Washington Hospital Center and MedStar Union Memorial Hospital in Baltimore, Md., offer WATCHMAN therapy to lessen the risk of AFib-related stroke without the need for long-term blood thinning medications.
“WATCHMAN technology has been shown to be a safe and effective alternative to blood-thinning medications in patients with AFib that is not caused by a heart valve problem. It is particularly valuable to patients who have a reason to avoid blood thinners,” said Cardiac Electrophysiologist Manish Shah, MD, director of the Cardiac Electrophysiology Fellowship Training Program at MedStar Washington Hospital Center. “We are honored to have implanted the 700th device at MedStar Health, and to offer patients this potentially life-improving therapy to reduce stroke risk. The field is advancing rapidly, with the new generation WATCHMAN FLX™ device, for example, and more innovations to come which will enable us to help even broader groups of patients.”
AFib is a major global health problem with as many as six million affected Americans. The condition is more common in older adults and results in an irregular heartbeat. Symptoms may or may not be obvious in patients but having AFib substantially increases the likelihood of stroke—on average, by five-fold.
The WATCHMAN implant closes off the left atrial appendage (LAA), an area of the heart where blood clots can form in patients with AFib. The device prevents harmful, potentially stroke-causing blood clots from forming and/or entering the bloodstream. This reduces stroke risk and enables patients to stop taking blood-thinning medications. The newest version of the device, WATCHMAN FLX, has an updated design to treat more patients safely and effectively, promoting the best long-term outcomes.
 “When we started implanting the WATCHMAN in 2016 at MedStar Union Memorial, there were certain criteria that had to be met to qualify a patient for the procedure. The technology has continued to improve since then, with five sizes available now, allowing us to treat very small and very large left atrial appendages. It’s been very gratifying to be on the forefront of this technology, that is both life-saving and life-changing,” said John Wang, MD, chief of the Cardiac Catheterization Laboratory at MedStar Union Memorial Hospital.
“When we started implanting the WATCHMAN in 2016 at MedStar Union Memorial, there were certain criteria that had to be met to qualify a patient for the procedure. The technology has continued to improve since then, with five sizes available now, allowing us to treat very small and very large left atrial appendages. It’s been very gratifying to be on the forefront of this technology, that is both life-saving and life-changing,” said John Wang, MD, chief of the Cardiac Catheterization Laboratory at MedStar Union Memorial Hospital.
WATCHMAN devices have been implanted in more than 150,000 patients worldwide. They are permanent devices that do not need replacement and cannot be seen outside the body. The procedure is performed in about an hour under general anesthesia. Patients spend one night in the hospital and are typically discharged the next morning. In 2015, MedStar Washington Hospital Center was the first hospital in the national capital region to implant the WATCHMAN device successfully.
Categories
Media Contact
So Young Pak
202-877-2748
soyoung.pak@medstar.net
Debra Schindler
410-274-1260
debra.schindler@medstar.net









