(Standing, l to r) Brian D. Baturin, MD; Preetham Kumar, MD; Ammar S. Bafi, MD; Ron Waksman, MD; Itsik Ben-Dor, MD; Christian C. Shults, MD; Toby Rogers, MD; Jeffrey E. Cohen, MD; (Seated, l to r) Gaby Weissman, MD; Lowell Satler, MD; Thomas E. MacGillivray, MD
Leading the way in correcting heart defects
Our structural heart and valvular heart disease experts are proud to offer patients access to the industry’s most advanced treatments, including the latest minimally invasive procedures, surgeries, and medical therapies.
Structural heart and valvular disease includes a variety of heart defects that a person can be born with or can acquire over time. These conditions include holes between the chambers of the heart, diseases of the heart muscles, or leaking or narrowing of the heart valves.
Patients travel from across the country to seek treatment from our specialists, particularly if they:
-
Cannot safely undergo traditional surgery
-
Have advanced disease or severe heart defects
-
Have co-morbid conditions
-
Require a procedure to supplement or correct a previous surgery
(Standing, l to r) John V. Conte, MD; Antony Kaliyadan, MD; Rachel E. Harrison, MD; (Seated, l to r) John Wang, MD; Brian T. Bethea, MD
We combine the expertise of cardiologists, cardiac surgeons, interventional cardiologists, and cardiovascular imaging specialists to treat patients with a spectrum of structural heart and valvular diseases at multiple sites in Virginia, Maryland, and the District of Columbia. These experts develop customized treatment plans for each patient, and the team meets daily to discuss and analyze all patients who are screened for any structural heart procedure.
Some of the many procedures our nationally recognized experts offer include:
Developing and testing new treatment options for heart and valve diseases
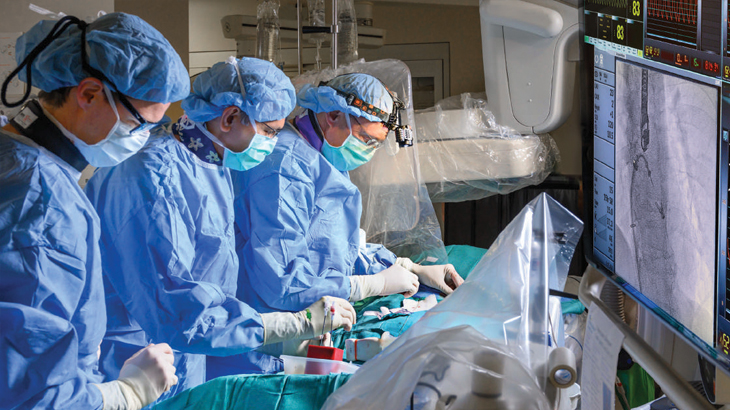
Our team is dedicated to advancing clinical research and new treatment options in this evolving field. We participate in a number of multicenter clinical trials, offering patients the chance to enroll in ongoing studies as treatment options.
We’re proud to have helped pioneer treatment and research in areas such as:
-
Tendyne: We are one of six centers evaluating this transcatheter-based mitral valve repair system that approaches its target through a small opening in the bottom of the heart.
-
Transcathether aortic valve replacement (TAVR): We have been a site for every major clinical trial for this procedure to treat aortic stenosis since the initial 2007 study. TAVR is currently approved by the Food and Drug Administration (FDA) for use in patients at high or intermediate risk of complications during traditional open-heart surgery. We are now evaluating its use in low-risk patients.
-
WATCHMAN™ device: Our specialists spent eight years testing this device to prevent stroke in patients with atrial fibrillation (AFib) before it reached the market in 2015. We’ve performed more of these procedures than any other heart center in the mid-Atlantic region.
Our providers
Location: Change location Enter your location
Structural Heart Disease Cardiology
-
Jennifer Rebecca Brown, MD
Cardiology, Structural Heart Disease Cardiology, Cardiovascular Disease & Heart Failure And Transplantation Cardiology
-
Erika D Feller, MD
Heart Failure And Transplantation Cardiology, Cardiovascular Disease, Advanced Heart Failure And Transplant Cardiology, Cardiology & Structural Heart Disease Cardiology
-
Shital Gandhi, MS PAC
Structural Heart Disease Cardiology, Heart Failure And Transplantation Cardiology & Cardiology
-
Rachel E Harrison, MD
Cardiac Surgery, Structural Heart Disease Cardiology, Thoracic Surgery & General Surgery
-
Ahmed N Khan, MD
Cardiology, Advanced Heart Failure And Transplant Cardiology, Cardiovascular Disease, Structural Heart Disease Cardiology & Heart Failure And Transplantation Cardiology
Insurance
MedStar Health accepts most major health insurance plans. If you are uncertain as to whether your individual health insurance plan is accepted at MedStar Health, please call your insurance company.










