Featured News
-
April 07, 2026
Carrington designed the colorful pants himself to give back to frontline healthcare workers in both DC and his hometown of Baltimore. WASHINGTON — By the time the first boxes were opened, excite...
Browse news by hospital and team
For reporters
Share this
All News
-
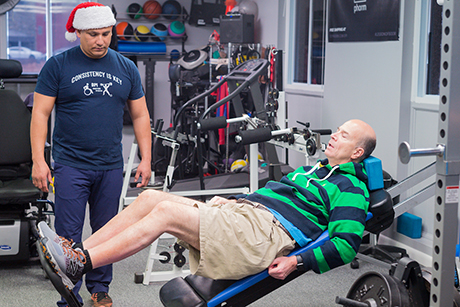 April 25, 2018
April 25, 2018Imagine living with a spinal cord injury as a result of simply falling in your home? The thought is quite scary and could happen to any one of us. And so it happened to Kent Keyser, who spent more t...
-
 April 23, 2018
April 23, 2018At age 13 Erik Weihenmayer lost his eyesight due to a condition called retinoschsis, characterized by the abnormal splitting of the retina’s neurosensory layers. Determined not to let his blindness b...
-
April 23, 2018
What causes a migraine? Migraines have a strong genetic component with 70 percent of patients having a first degree relative with a history of migraines. More women than men suffer migraines, a...
-
 April 18, 2018
April 18, 2018Washington, D.C., April 18, 2018 – Clinical trial planning is underway at MedStar Heart & Vascular Institute to determine whether a novel stem cell therapy will improve heart function for patients...
-
April 17, 2018
Mary Hart, second from right, was recently named the 2018 Auxilian of the Year. Hart stands with MedStar St. Mary’s Hospital President Christine Wray, right, close friend Ernestine Pence, and daughter...
-
April 17, 2018
Neurologist Aabha Shah, MD, with patient Adrian Knight. A trip to Golden Corral began like any other lunch for Adrian Knight, a trim, previously healthy, retired IRS worker, and father of two grown d...
- 1
- 2
- 3
- 4
- 5
- 6
- 7
- 8
- 9
- 10
- 11
- 12
- 13
- 14
- 15
- 16
- 17
- 18
- 19
- 20
- 21
- 22
- 23
- 24
- 25
- 26
- 27
- 28
- 29
- 30
- 31
- 32
- 33
- 34
- 35
- 36
- 37
- 38
- 39
- 40
- 41
- 42
- 43
- 44
- 45
- 46
- 47
- 48
- 49
- 50
- 51
- 52
- 53
- 54
- 55
- 56
- 57
- 58
- 59
- 60
- 61
- 62
- 63
- 64
- 65
- 66
- 67
- 68
- 69
- 70
- 71
- 72
- 73
- 74
- 75
- 76
- 77
- 78
- 79
- 80
- 81
- 82
- 83
- 84
- 85
- 86
- 87
- 88
- 89
- 90
- 91
- 92
- 93
- 94
- 95
- 96
- 97
- 98
- 99
- 100
- 101
- 102
- 103
- 104
- 105
- 106
- 107
- 108
- 109
- 110
- 111
- 112
- 113
- 114
- 115
- 116
- 117
- 118
- 119
- 120
- 121
- 122
- 123
- 124
- 125
- 126
- 127
- 128
- 129
- 130
- 131
- 132
- 133
- 134
- 135
- 136
- 137
- 138
- 139
- 140
- 141
- 142
- 143
- 144
- 145
- 146
- 147
- 148
- 149
- 150
- 151
- 152
- 153
- 154
- 155
- 156
- 157
- 158
- 159
- 160
- 161
- 162
- 163
- 164
- 165
- 166
- 167
- 168
- 169
- 170
- 171
- 172
- 173
- 174










