Featured Blog
-
 May 26, 2026
May 26, 2026Our research, published in the Journal of Cancer Survivorship, helps us understand the risk of functional limitations after cancer treatment, enabling us to improve care for survivors. There are ma...
Browse blogs by hospital
Share this
All Blogs
-
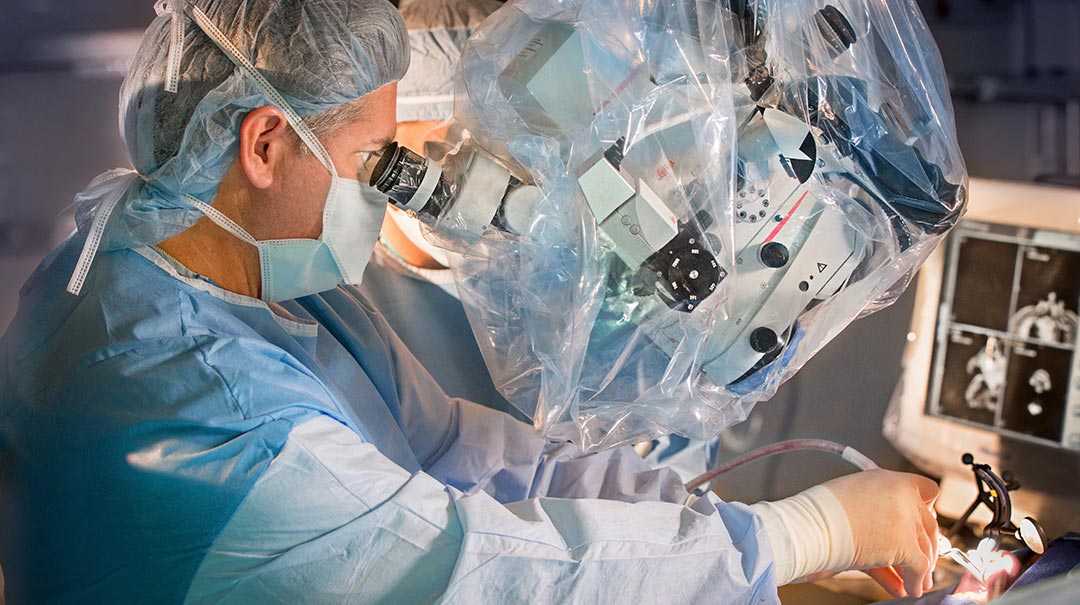 April 04, 2019
April 04, 2019Imagine this scenario: You come to the hospital with a complex neurological condition, and your surgeon uses a device or instrument you’ve never seen before—a tool that is perfectly designed to approa...
-
 April 04, 2019
April 04, 2019By MedStar Health Research Institute
Informed Consent for Clinical Research The Informed Consent Form (ICF) template at MHRI was updated Spring 2018. As of January 21, 2019 this template is required for all new submissions being reviewe...
-
 April 02, 2019
April 02, 2019Patients often turn to corrective jaw surgery to correct irregularities in their jaw and teeth to improve chewing, speaking, and appearance. However, an additional condition that many people don’t rea...
-
 March 28, 2019
March 28, 2019Drinking diet soda might seem like a good idea, as it contains zero calories. However, drinking two or more artificially sweetened drinks a day could significantly increase the risk of heart disease, ...
-
 March 26, 2019
March 26, 2019By MedStar Team
The techniques used to diagnose and treat colorectal cancer have become better each and every year—in fact, many of them didn’t exist or were in early research stages 10 to 20 years ago. ...
-
 March 21, 2019
March 21, 2019When someone comes into the emergency department (ED) with major burns over a large area of their body, it’s obvious that they need immediate medical attention. However, smoke inhalation—...
- 1
- 2
- 3
- 4
- 5
- 6
- 7
- 8
- 9
- 10
- 11
- 12
- 13
- 14
- 15
- 16
- 17
- 18
- 19
- 20
- 21
- 22
- 23
- 24
- 25
- 26
- 27
- 28
- 29
- 30
- 31
- 32
- 33
- 34
- 35
- 36
- 37
- 38
- 39
- 40
- 41
- 42
- 43
- 44
- 45
- 46
- 47
- 48
- 49
- 50
- 51
- 52
- 53
- 54
- 55
- 56
- 57
- 58
- 59
- 60
- 61
- 62
- 63
- 64
- 65
- 66
- 67
- 68
- 69
- 70
- 71
- 72
- 73
- 74
- 75
- 76
- 77
- 78
- 79
- 80
- 81
- 82
- 83
- 84
- 85
- 86
- 87
- 88
- 89
- 90
- 91
- 92
- 93
- 94
- 95
- 96
- 97
- 98
- 99
- 100
- 101
- 102
- 103
- 104
- 105
- 106
- 107
- 108
- 109
- 110
- 111
- 112
- 113
- 114
- 115
- 116
- 117
- 118
- 119
- 120
- 121
- 122
- 123
- 124
- 125
- 126
- 127
- 128
- 129
- 130
- 131
- 132
- 133
- 134
- 135
- 136
- 137
- 138
- 139
- 140
- 141
- 142
- 143
- 144
- 145
- 146
- 147
- 148
- 149
- 150
- 151
- 152
- 153
- 154
- 155
- 156
- 157
- 158
- 159
- 160
- 161
- 162
- 163
- 164
- 165
- 166
- 167
- 168
- 169
- 170
- 171
- 172
- 173
- 174
- 175
- 176
- 177
- 178
- 179
- 180
- 181
- 182
- 183
- 184
- 185
- 186
- 187
- 188
- 189
- 190
- 191
- 192
- 193
- 194
- 195
- 196
- 197
- 198
- 199
- 200
- 201
- 202
- 203
- 204
- 205
- 206
- 207
- 208
- 209
- 210
- 211
- 212
- 213
- 214
- 215
- 216
- 217
- 218
- 219
- 220
- 221
- 222
- 223
- 224
- 225
- 226
- 227
- 228
- 229
- 230
- 231
- 232
- 233
- 234
- 235
- 236
- 237
- 238
- 239
- 240
- 241
- 242
- 243
- 244
- 245
- 246
- 247
- 248
- 249
- 250
- 251
- 252
- 253
- 254
- 255
- 256
- 257
- 258
- 259
- 260
- 261
- 262
- 263
- 264
- 265
- 266
- 267
- 268
- 269
- 270
- 271
- 272
- 273
- 274
- 275
- 276
- 277
- 278
- 279
- 280
- 281
- 282
- 283
- 284
- 285
- 286
- 287
- 288
- 289
- 290
- 291
- 292
- 293
- 294
- 295
- 296
- 297
- 298
- 299
- 300
- 301










